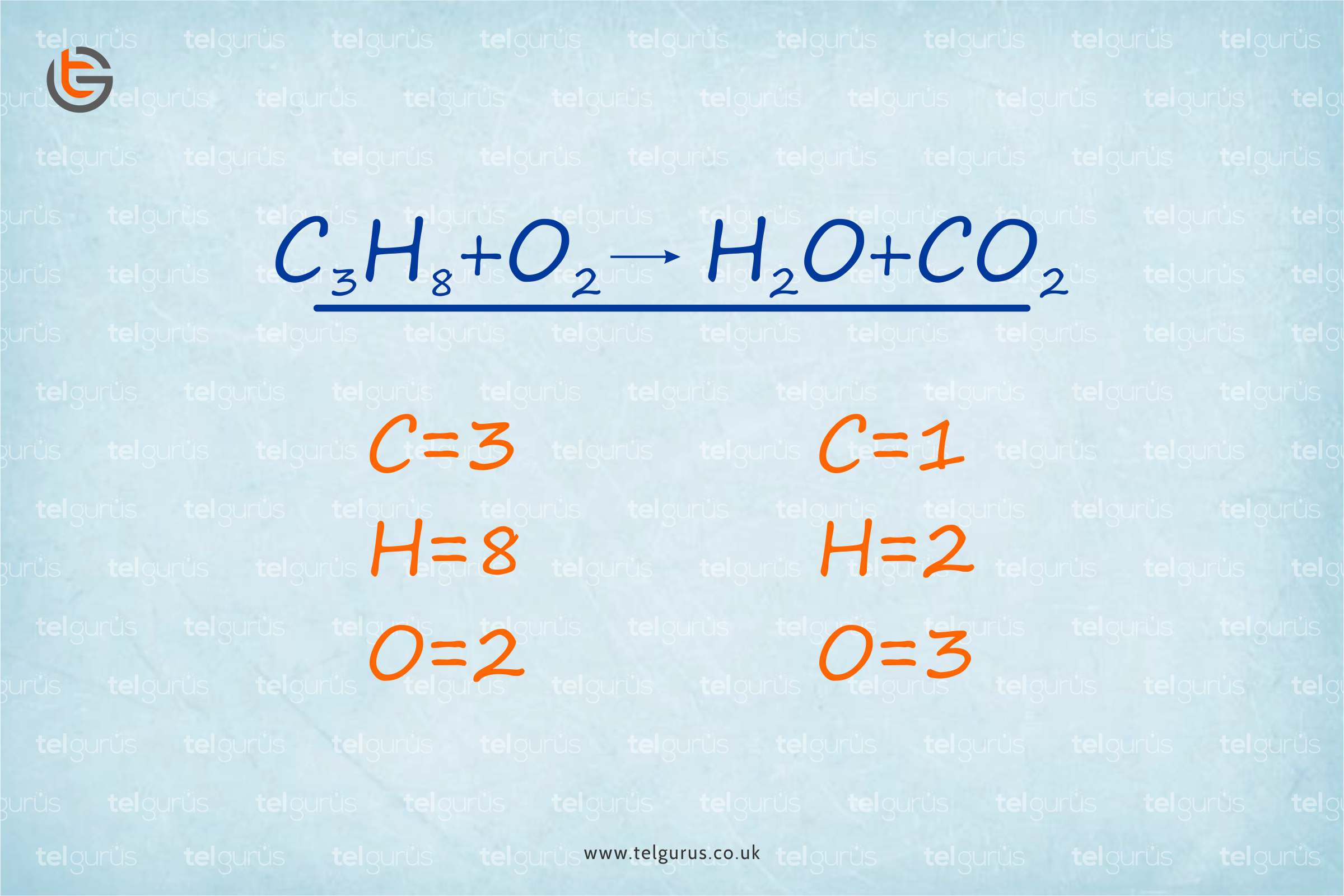 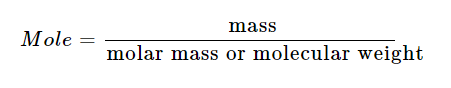 
Notice that in these two examples N 2 and O 2 react with a different stoichiometry to obtain different products. However, we can get rid of the fractional coefficient by again multiplying by 2. There are already 2 atoms of N so we can add 3/2 in front of oxygen Number of atoms we need 2 atoms of N and 3 atoms of oxygen on the left side of Now we can balance the remaining single element species. Is the most complex species so we will add a 1 for its coefficient. This one may not be as easy to see the final answer so we will use the guidelines to balance the equation. You can seeīy simply adding a 2 in front of NO, we violate the first guideline even though Remember they are only guidelines to help if you run into trouble. If you can see the balanced equation by sight, you don't need to go by the guidelines. This is a perfectly acceptable balanced chemical equation.Īt the very beginning of this problem, perhaps you could see this was the answer. We can get rid of the fractional coefficients by multiplying by 2 even though Is not explicitly included when writing the chemical equation. Note: Typically a stoichiometric coefficient of "1" This we will need to use fractional coefficients. Now we can balance the remaining single-element compounds. Assign a stoichiometricĬoefficient of 1 to the most complex compound, NO. This equation is not balanced since there are more N and O atoms on the left Therefore, sometimes it may be necessary to deviate from these general There must be the same number of atoms on the left and right sides.Add coefficients only do not change the chemical formulas.Eliminate fractional coefficients (although this is not necessary).Balance any single-element species last.Assign a "1" as the coefficient for the most complex species (the one whose chemical formula has the greatest number of different elements).General Guidelines for Balancing Simple Equations These coefficients are the stoichiometricĬoefficients. We balance reactions by addingĬoefficients in front of the reactants and products. You see a chemical reaction is to balance it. If a chemical reaction is not balanced, no information about the relationshipīetween products and reactants can be derived. Of various atoms in the products as in the reactants. Products and reactants in a chemical reaction.Ĭhemical reactions must be balanced, or in other words, must have the same number Quantitative relation between the number of moles (and therefore mass) of various In chemistry it is very important to understand the relationship between reactantsĪnd products in a reaction. Overview: In this tutorial, the fundamentals of balancing chemical
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
